Hydrothermal Synthesis Of Nanomaterials Pdf
Nanomaterials: Synthesis, Properties and Applications, Second Edition 2nd Edition. ISBN-13: 978-0750305785. Chapter 2.10: Further Methods for the Synthesis of Nanomaterials. Nanostructures & Nanomaterials: Synthesis, Properties & Applications . Nanomaterials: Synthesis, Properties and Applications Edited by A S Edelstein Naval Research Laboratory Washington, DC and R
Nanomaterials - Wikipedia, the free encyclopedia. See the Nanomaterials category for an exhaustive list of articles related to this subject. What follows is an introduction.
Chapter - INTRODUCTION TO NANOMATERIALS A. Introduction Nanomaterials are cornerstones of nanoscience and nanotechnology. Nanomaterial - synthesis and processing Nanomaterials deal with very fine structures.
Solution Combustion Synthesis Of Nanomaterials Pdf
Nanomaterials describe, in principle, materials of which a single unit is sized (in at least one dimension) between 1 and 1. Materials with structure at the nanoscale often have unique optical, electronic, or mechanical properties. Nanomaterials are slowly becoming commercialized. The structure of foraminifera (mainly chalk) and viruses (protein, capsid), the wax crystals covering a lotus or nasturtium leaf, spider and spider- mite silk. For example, clays display complex nanostructures due to anisotropy of their underlying crystal structure, and volcanic activity can give rise to opals, which are an instance of a naturally occurring photonic crystals due to their nanoscale structure.
Fires represent particularly complex reactions and can produce pigments, cement, fumed silica etc. The play of color is caused by the interference and diffraction of light between silica spheres (1. Lycurgus Cup, glass, 4th century, Roman. Nanoparticles (7. Blue hue of a species of tarantula (4. These include the carbon nanotubes (or silicon nanotubes) which are of interest both because of their mechanical strength and also because of their electrical properties. The name was a homage to Buckminster Fuller, whose geodesic domes it resembles.
Fullerenes have since been found to occur in nature. In April 2. 00. 3, fullerenes were under study for potential medicinal use: binding specific antibiotics to the structure of resistant bacteria and even target certain types of cancer cells such as melanoma. The October 2. 00. Chemistry and Biology contains an article describing the use of fullerenes as light- activated antimicrobial agents. In the field of nanotechnology, heat resistance and superconductivity are among the properties attracting intense research.
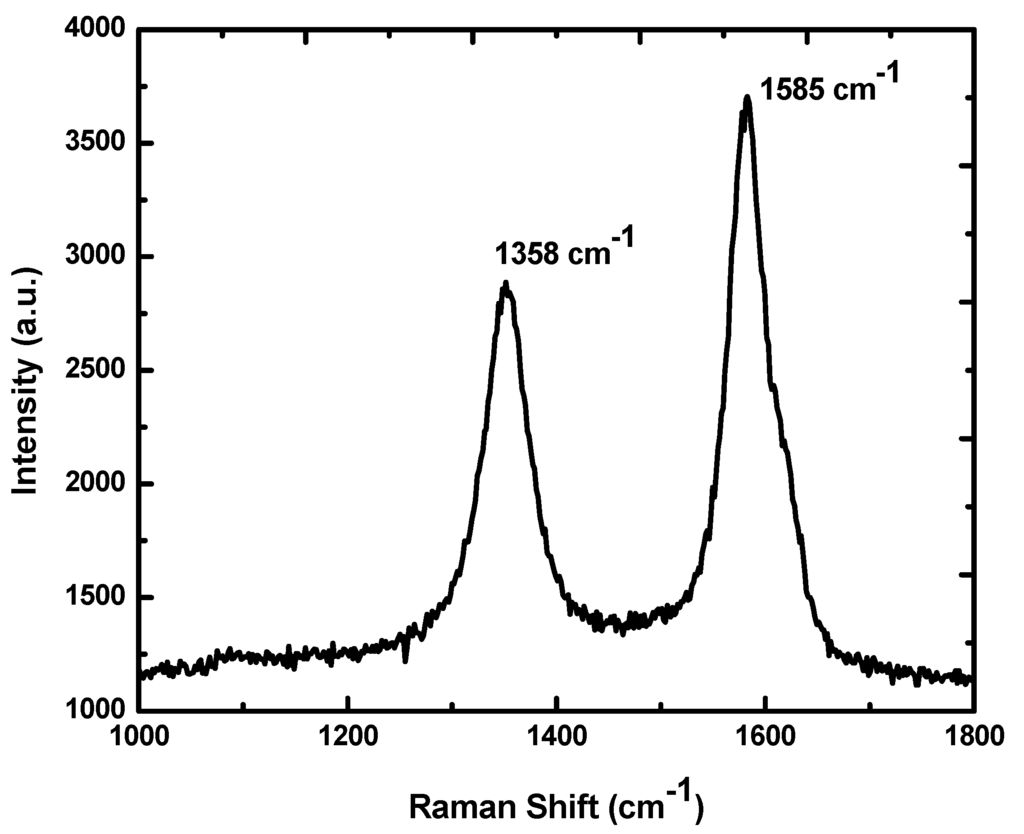
A common method used to produce fullerenes is to send a large current between two nearby graphite electrodes in an inert atmosphere. The resulting carbonplasma arc between the electrodes cools into sooty residue from which many fullerenes can be isolated. There are many calculations that have been done using ab- initio Quantum Methods applied to fullerenes. By DFT and TDDFT methods one can obtain IR, Raman and UV spectra. Results of such calculations can be compared with experimental results. Graphene nanostructures. The most important representative graphene was discovered in 2.
Other 2. D materials based on other elements have since been reported. Box- shaped graphene (BSG) nanostructure is an example of 3. D nanomaterial. This nanostructure is a multilayer system of parallel hollow nanochannels located along the surface and having quadrangular cross- section. The thickness of the channel walls is approximately equal to 1 nm.
The typical width of channel facets makes about 2. Nanoparticles. There are the possibilities to use those materials in organic material based optoelectronic devices such as Organic solar cells, OLEDs etc.
The operating principles of such devices are governed by photoinduced processes like electron transfer and energy transfer. The performance of the devices depends on the efficiency of the photoinduced process responsible for their functioning. Therefore, better understanding of those photoinduced processes in organic/inorganic nanomaterial composite systems is necessary in order to use them in organic optoelectronic devices. Nanoparticles or nanocrystals made of metals, semiconductors, or oxides are of particular interest for their mechanical, electrical, magnetic, optical, chemical and other properties. Nanoparticles have been used as quantum dots and as chemical catalysts such as nanomaterial- based catalysts.
Recently, a range of nanoparticles are extensively investigated for biomedical applications including tissue engineering, drug delivery, biosensor. A bulk material should have constant physical properties regardless of its size, but at the nano- scale this is often not the case. Size- dependent properties are observed such as quantum confinement in semiconductor particles, surface plasmon resonance in some metal particles and superparamagnetism in magnetic materials.
Nanoparticles exhibit a number of special properties relative to bulk material. For example, the bending of bulk copper (wire, ribbon, etc.) occurs with movement of copper atoms/clusters at about the 5.
Copper nanoparticles smaller than 5. The change in properties is not always desirable. Ferroelectric materials smaller than 1. Suspensions of nanoparticles are possible because the interaction of the particle surface with the solvent is strong enough to overcome differences in density, which usually result in a material either sinking or floating in a liquid. Nanoparticles often have unexpected visual properties because they are small enough to confine their electrons and produce quantum effects. For example, gold nanoparticles appear deep red to black in solution.
The often very high surface area to volume ratio of nanoparticles provides a tremendous driving force for diffusion, especially at elevated temperatures. Sintering is possible at lower temperatures and over shorter durations than for larger particles. This theoretically does not affect the density of the final product, though flow difficulties and the tendency of nanoparticles to agglomerate do complicate matters. The surface effects of nanoparticles also reduces the incipient melting temperature. Nanozymes. Accordingly, the synthetic method should exhibit control of size in this range so that one property or another can be attained.
Often the methods are divided into two main types . In these methods the raw material sources can be in the form of gases, liquids or solids. The latter requiring some sort of disassembly prior to their incorporation onto a nanostructure. Bottom methods generally fall into two categories: chaotic and controlled. Chaotic processes. Through the clever manipulation of any number of parameters, products form largely as a result of the insuring kinetics. The collapse from the chaotic state can be difficult or impossible to control and so ensemble statistics often govern the resulting size distribution and average size.
Accordingly, nanoparticle formation is controlled through manipulation of the end state of the products. Examples of Chaotic Processes are: Laser ablation, Exploding wire, Arc, Flame pyrolysis, Combustion, Precipitation synthesis techniques. Controlled processes. Generally the state of the constituent atoms or molecules are never far from that needed for nanoparticle formation.
Accordingly, nanoparticle formation is controlled through the control of the state of the reactants. Examples of controlled processes are self- limiting growth solution, self- limited chemical vapor deposition, shaped pulse femtosecond laser techniques, and molecular beam epitaxy. Characterization. In these cases quantum mechanical effects can dominate material properties. One example is quantum confinement where the electronic properties of solids are altered with great reductions in particle size. The optical properties of nanoparticles, e. This effect does not come into play by going from macrosocopic to micrometer dimensions, but becomes pronounced when the nanometer scale is reached.
In addition to optical and electronic properties, the novel mechanical properties of many nanomaterials is the subject of nanomechanics research. When added to a bulk material, nanoparticles can strongly influence the mechanical properties of the material, such as the stiffness or elasticity. For example, traditional polymers can be reinforced by nanoparticles (such as carbon nanotubes) resulting in novel materials which can be used as lightweight replacements for metals. Such composite materials may enable a weight reduction accompanied by an increase in stability and improved functionality. The further development of such catalysts can form the basis of more efficient, environmentally friendly chemical processes. The first observations and size measurements of nano- particles were made during the first decade of the 2.
Zsigmondy made detailed studies of gold sols and other nanomaterials with sizes down to 1. He published a book in 1. These are widely used for first generation passive nanomaterials specified in the next section. These methods include several different techniques for characterizing particle size distribution. This characterization is imperative because many materials that are expected to be nano- sized are actually aggregated in solutions. Some of methods are based on light scattering.
Others apply ultrasound, such as ultrasound attenuation spectroscopy for testing concentrated nano- dispersions and microemulsions. This information is required for proper system stabilzation, preventing its aggregation or flocculation. These methods include microelectrophoresis, electrophoretic light scattering and electroacoustics. The last one, for instance colloid vibration current method is suitable for characterizing concentrated systems. Uniformity. In condensed bodies formed from fine powders, the irregular sizes and shapes of nanoparticles in a typical powder often lead to non- uniform packing morphologies that result in packing density variations in the powder compact. Uncontrolled agglomeration of powders due to attractivevan der Waals forces can also give rise to in microstructural inhomogeneities.
Differential stresses that develop as a result of non- uniform drying shrinkage are directly related to the rate at which the solvent can be removed, and thus highly dependent upon the distribution of porosity. Such stresses have been associated with a plastic- to- brittle transition in consolidated bodies, and can yield to crack propagation in the unfired body if not relieved.
 RSS Feed
RSS Feed
